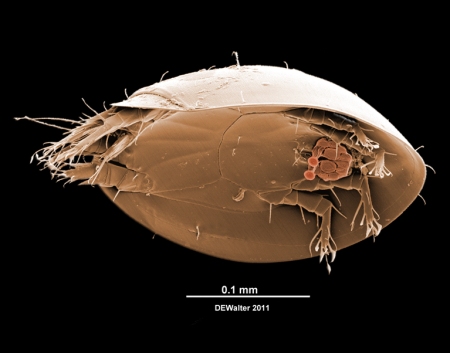
This is the first Challenge using a light micrograph, but the usual rules apply – name the beasts as well as you can and Macromite fame may be yours. If the task proves too daunting, well then here’s a meander through the illustrated behavior to keep you entertained.
Phoresy is a behaviour, or rather a set of behaviours, exhibited by many small animals and especially by mites. The word is a bit confusing. The root, phor– is from the Greek word phoros which means to bear, to carry or refers to movement (not phor, a thief or kind of bee), but phoresy actually requires an absence of movement and it is usually not (but not always) the mite that does the carrying. Thus, the usage is a bit inverted from similarly derived jargon, e.g. the conidiophores that bear fungal spores or electrophoresis where an electrical field is used to carry charged particles through a gel or other medium. In the case of phoresy, it is the mite that is carried, usually by some lucky, larger, and more vagile arthropod.
Nature is full of interesting interactions, but to study them first requires defining them. Well, perhaps not, but that is what scientists like to do – and tying them up with definitions and tagging them with a name derived from Greek or Latin (or both) is always great fun. The term phoresy was first proposed by French entomologist and bostrichid specialist Pierre Lesne in 1896 and to quote from a delightful paper (read the pdf – the web text is full of errors) by the famous myrmecophile and ant acarologist W.M. Wheeler (1919): “In 1896 Lesne called attention to a number of small insects that habitually ride on larger insects. To this phenomenon he applied the term “phoresy” and showed that it is distinguished from ectoparasitism by the fact that the portee does not feed on the porter and eventually dismounts and has no further relations with the latter.”
As classically defined in the mid-20th Century, phoresy required an animal to stop its normal behaviours (e.g. seeking food or members of the opposite sex), seek out a carrier, mount the carrier and refrain from doing anything other than holding on, and then after a time to dismount from the carrier and resume normal behaviours. Taking a bite out of the carrier was considered a no-no – and animals that did so were banned from phoresy and sent to parasite prison. The heteromorphic deutonymph of Astigmatina (aka hypopus) is a classic phoriont – it lacks functional mouthparts, has no foregut (and so feeding would seem to be verboten), and has a ventral sucker disk formed from modified setae for holding onto an insect or other larger arthropod (sometimes even other mites).
So, in an evolutionary ecology sense, classical phoresy can be thought of as an ectosymbiosis in the commensal category where the mite gets a free ride and the carrier doesn’t really care. Dispersal is assumed to be at the root of the evolution of these behaviours in the mites (although phoresy may actually concentrate, rather than disperse the mites). But are things really so simple?
Again the jargon is a bit confusing: ‘commensal’ implies eating together, and the mite can’t eat while on the carrier or it wouldn’t be considered phoretic. Consider the case of water mite larvae – they use adult aquatic insects to ‘disperse’, but since they take a bite while doing so, they are considered parasites, not phorionts. Then there are the strange hypopi of Hemisarcoptes cooremani which hitch rides under the elytra of ladybird beetles in the genus Chilochorus. Since Chilochorus beetles like to eat mites, this seems like a dangerous thing to do – but hypopi of H. cooremani that do not find a beetle die. Those that do find a beetle hang on for 5-21 days and swell up before leaving the beetle. Marilyn Houck (1994), who has studied this interaction, has suggested that beetle hemolymph (reflexive bleeding is a defense of many ladybird beetles – pick them up and noxious yellow blood oozes from their leg joints) may provide critical nutrients, possibly via the anal vestibule of the mites (which connects to a hindgut).
Another point to consider is how the porter really feels about the portees. When I look at insects covered in phoretic mites, e.g. the Photon Challenge above, I find it difficult to believe the bug is having a good time. But some bees and wasps have special mite pockets (acarinaria) that appear to have evolved to encourage mites to hitch a ride. I’ll post on acarinaria some day, but for now I will just note that some of these hymenopters do much better with their mites than without them.
Lisa Hodgkin and her colleagues at the University of Melbourne published an excellent paper last year with some interesting experimental data. They studied a bark beetle (Ips grandicollis) introduced into Australia and its phoretic mites and demonstrated that ‘phoresy’ can be both dynamic and complex. Phoretic mites were associated with both negative (adults) and positive (larvae) effects on beetle reproduction and development. Heavy mite loads were not good for female beetles, but having mites in the galleries resulted in bigger and healthier offspring. Perhaps when we are considering defining complex interactions like phoresy we should remember what Hamlet pointed out to Horatio: There are more things in heaven and earth, Horatio, Than are dreamt of in your philosophy.
References:
Lisa K. Hodgkin, Mark A. Elgar and Matthew R. E. Symonds. 2010. Positive and negative effects of phoretic mites on the reproductive output of an invasive bark beetle. Australian Journal of Zoology, 2010, 58, 198–204. http://www.publish.csiro.au/paper/ZO10034
Houck, M.A. 1994. Adaptation and transition into parasitism from commensalism: a phoretic model. In: (Houck, M.A., ed.), Mites. Ecological and evolutionary analyses of life-history patterns. Chapman and Hall, New York: 252-281.
P. Lesne. 1896. Moeurs du Limosina sacra. Phenomenes de transport mutuel chez les animaux articules, Origine du parasitisme chez les insectes Dipteres. Bull. Soc. Ent. France 45, 1896, pp. 162-165.
WM Wheeler. 1919. The phoresy of Antherophagus. Psyche Boston Volume: 26: 145-152.
http://psyche.entclub.org/26/26-145.html